Research
Stable Monomolecular Interfaces
The goal of this project is to investigate an interfacial organic carbene insertion on hard and soft material interfaces. Our overarching aim is to develop a general molecular layer deposition technique, capable of functionalizing diverse materials (inorganic, organic, polymeric including flat, curved, rough or nanostructured interfaces) with uniform and stable monolayers of functional organic molecules. Instead of using self-assembly and ordering to form stable monolayers, we utilize diazirine molecules to enable solution or vapor-based carbenylation and rapid formation of thermodynamically and kinetically stable surface-monolayer bonds. Such chemisorption produces dense and uniform monolayers that do not require extensive intermolecular interactions for stability.
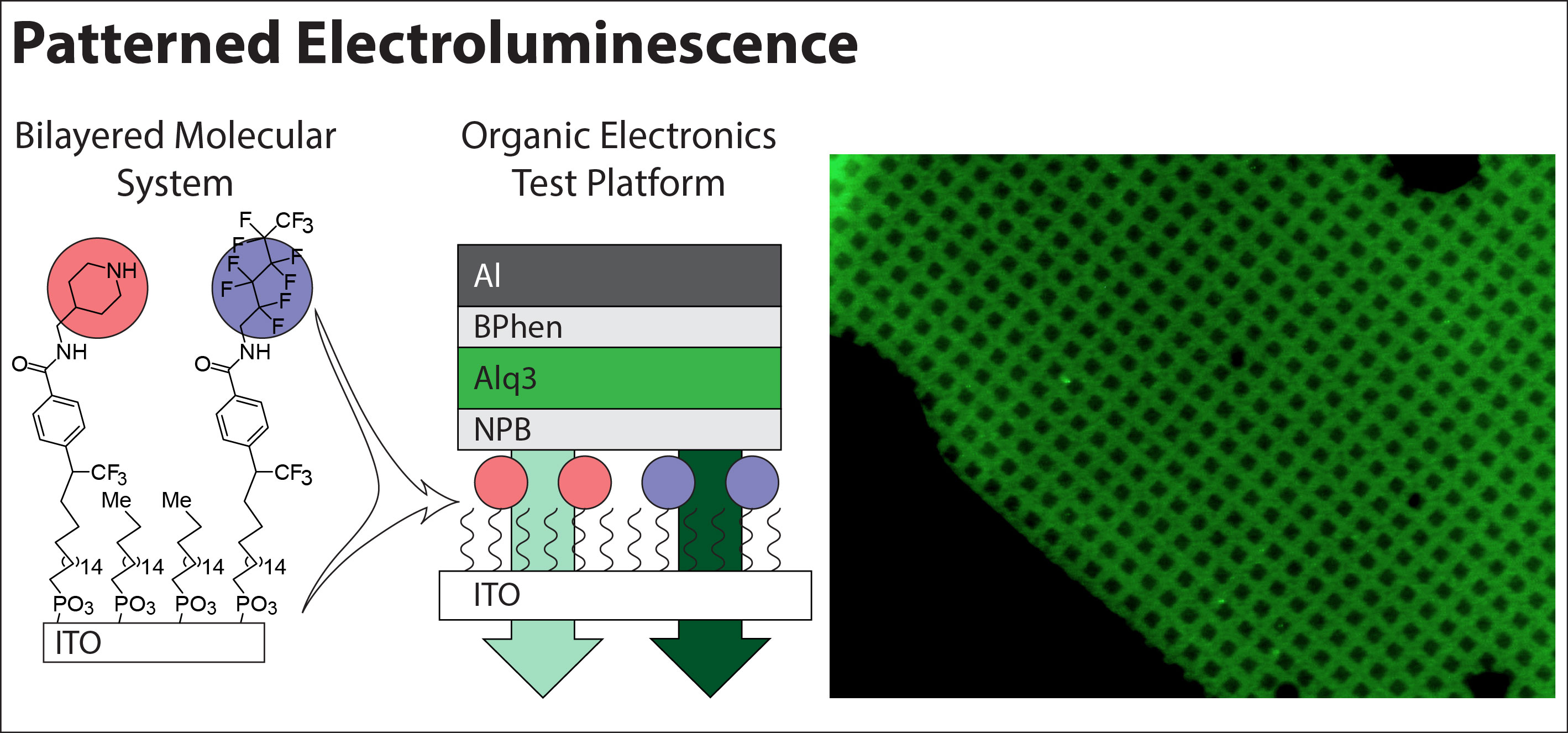
Within this project, we recently used a novel surface engineering approach to demonstrate the influence of organic functional group substitutions on molecular electronic properties. Specifically, we compared carbeynylated monolayers immobilized on an inorganic electrode as the charge-injecting components of organic electronic devices. Our carbenylated interfaces comprised inert and functional layers whose properties can be independently controlled from each other. We demonstrated that the charge transfer through our monolayers is sensitive to small structural molecular changes; that it can be controlled and predicted by controlling the electron-withdrawing or donating nature of the organic moiety; and that the differences in the charge transfer dynamics of two monolayers can be visualized via patterned electroluminescence.
We also demonstrated that functional diazirines can be used to modify methylated and Si-H terminated surfaces via vapor-phase UV-catalyzed carbene insertion. We demonstrated that methylated silicon, hydrogen-terminated silicon, hydrogen-terminated silicon nitride and inert polyurethane-acrylate polymer containing methylated molecules can be functionalized with the organic diazirines, which remain stable on inert interfaces even after a two-week exposure to water and various organic solvents.
Publications:
X Li, D Johnson, W Ma, H Chung, J Getpreecharsawas, JL McGrath, AA Shestopalov, Chemistry of Materials, 2016, submitted
X Li, W Ma, AA Shestopalov, Langmuir, 2016, DOI: 10.1021/acs.langmuir.6b02471
AA Shestopalov, JL McGrath, X Li, US Patent application number 15/130,208, 2016 (Pending, Filing Date: 04/15/2016)
FF Angel, Y Lyubarskaya, AA Shestopalov, CW Tang; Organic Electronics, 2014, 15, 3624-3631
CM Bowers, M Zhang, Y Lyubarskaya, CW Tang, AA Shestopalov; Advanced Materials Interfaces, 2014, 1(2), 1300109
DG Johnson, TS Khire, YL Lyubarskaya, KJP Smith, JP DesOrmeaux, JG Taylor, TR Gaborski, AA Shestopalov, CC Striemer, JL McGrath, Advances in Chronic Kidney Disease, 2013, 20(6), 508–515
CM Bowers, EJ Toone, RL Clark, AA Shestopalov, Journal of Visualized Experiments, 2011, 58, 1–7
Support:
NSF DMR 1228889
UR PumpPrimer