New imaging technology could buy time for pancreatic cancer patients
Tumor shrinkage is one sign of cancer treatment’s efficacy—but Rochester scientists are exploring elasticity and permeability as well.

The insidiousness of pancreatic cancer is how it develops without showing any definitive symptoms. In most cases, by the time it is diagnosed, it is beyond cure.
And yet, for 10 to 20 percent of patients, pancreatic cancer is caught soon enough, before it has metastasized. This provides surgeons a narrow window of time to try to treat the tumors, shrinking them enough to safely remove them.
University of Rochester engineers, imaging scientists, surgeons, and immunologists are working together on a novel imaging technology to help surgeons make the most of that narrow time frame before the cancer spreads.
Led by Marvin Doyley, professor and chair of electrical and computer engineering at the Hajim School of Engineering and Applied Sciences, the collaborators have received a $2.4 million grant from the National Institute of Biomedical Imaging and Bioengineering to look at pancreatic cancer tumors in a different way—measuring not just their size, but also their elasticity and ability to be perfused, or permeated, with blood and other fluids.
“Standard imaging modalities such as MRI (magnetic resonance imaging) or CT (computed tomography) have been used to see if a tumor is shrinking,” Doyley says. “But there is a lot of data that suggests just looking at size reduction by itself is not necessarily the best marker to judge whether patients are responding to therapy. We’re basically trying to fix that.”
The ultimate goal is to develop an endoscopic device that surgeons could use to look inside the body in order to determine whether a patient is responding to treatment and, if so, when they are ready to undergo tumor removal.
The first step is to demonstrate whether increases in tumor elasticity and perfusion are indeed valid biomarkers to more quickly determine that a tumor is responding to treatment in ways that will help ensure a successful surgical outcome. And if not, to switch to a different treatment.
Overcoming tumor barriers with two types of imaging
Pancreatic cancer tumors typically have a stiff barrier around them, which can pinch off the ability of blood vessels to perfuse the tumor with chemotherapy drugs designed to shrink the tumor, Doyley explains.
To address the specific challenges of pancreatic cancer tumors, the researchers will use a combination of two imaging modalities:
- Shear wave elastography, a type of ultrasound used to map the elastic properties and stiffness of tissues
- Optical fluorescence tomography, a type of optical imaging capable of precisely measuring perfusion in tiny vessels at the molecular level
This will enable the team to measure whether tumor elasticity and perfusion change in mice models as they respond to various tumor-shrinking therapies—and if so, to “fine tune” the ability of the imaging technology to detect the changes.
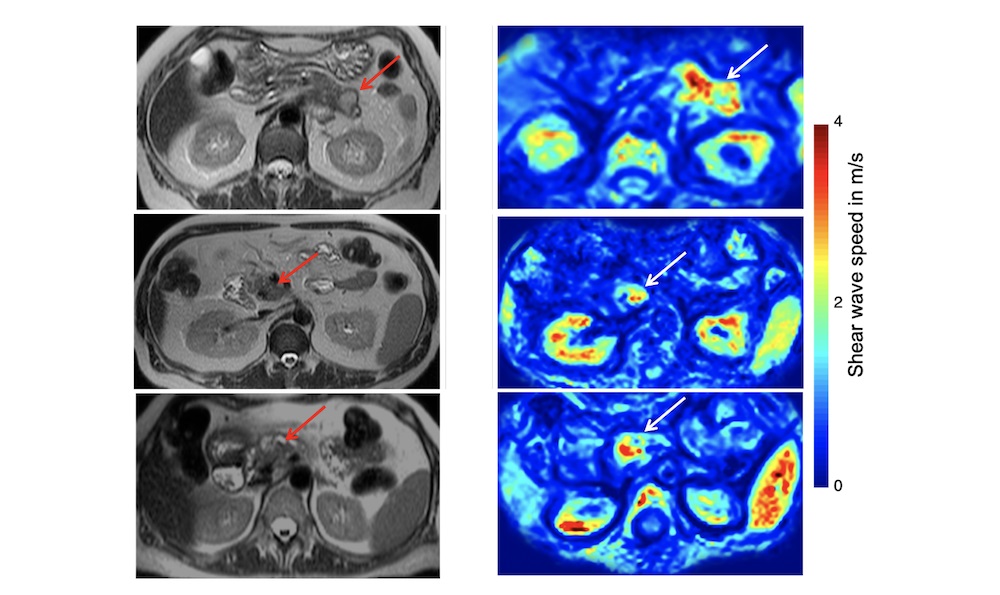
Then, the researchers will use analogous forms of MRI imaging to see how the same properties change in actual patients undergoing treatment.
“Once a patient is initially diagnosed with pancreatic cancer, we’ll track how those biomarkers vary as they have their different treatments,” Doyley says. “We can’t do a full clinical trial, but we can retrospectively see, in patients who have responded, if there is a corresponding change in the biomarkers we are looking at.”
And if there is indeed a link between the biomarkers and treatments, continues Doyley, “we will apply for a bigger grant to create an endoscopic device and do a bigger study with more patients.”
Collaborators include Brian Pogue, recently appointed chair of the Department of Medical Physics at the University of Wisconsin–Madison, and, at Rochester, David Lenihan, professor and chair of surgery; Scott Gerber, associate professor of surgery and of microbiology and immunology; Aram Hezel, associate professor of hematology/oncology; Tanzy Love, associate professor of biostatistics; Jonathan Kallas, assistant professor of imaging sciences, and David Dombroski, associate professor of imaging sciences.
