
Mark Buckley
Associate Professor of Biomedical Engineering
Center for Musculoskeletal Research
Center for Visual Science
PhD, Cornell University, 2010
- Office Location
- 317 Goergen Hall
- Telephone
- (585) 276-4195
- Web Address
- Website
Biography
Mark Buckley received his Ph.D. in Physics from Cornell University under the mentorship of Drs. Itai Cohen and Lawrence Bonassar in 2010 and worked under Dr. Louis Soslowsky as a post-doctoral fellow at the University of Pennsylvania from 2010-2012. He joined the faculty of the Department of Biomedical Engineering at the University of Rochester in January of 2013. He has co-authored publications on diverse topics ranging from three-dimensional tracking of swimming bacteria to the use of ultrasound elastography to quantify transverse compressive strain in the Achilles tendon. Dr. Buckley is particularly interested in the mechanics of soft biological tissues including cartilage, tendon and the cornea. His research emphasizes finding ways to control and exploit their complex mechanical properties to prevent graft damage during transplantation surgeries, diagnose injury and disease, guide rehabilitation protocols, and evaluate treatment and repair strategies.
Research Overview
The goal of the Buckley lab is to exploit the principles of mechanics to improve treatments for diseases and injuries affecting soft biological tissues. Currently, we are primarily focused on musculoskeletal and ocular tissues including cartilage, tendon, intervertebral disc and cornea, and we study diseases ranging from osteoarthritis to keratoconus.
For example, we are investigating novel strategies to reduce damage to transplanted grafts during osteochondral (cartilage with bone) and corneal transplantation surgery. In these procedures, accidental graft damage increases risk of graft failure, but may be avoidable. We have begun to determine the types (e.g., shear or tensile) and magnitudes of mechanical stress experienced by sensitive regions of cartilage and cornea during transplantation procedures, as well as the types and magnitudes of mechanical stress that cause the greatest extent of damage to these tissues and their resident cells. Based on our findings, we are designing devices that either prevent imposition of stresses exceeding a critical level or alter graft properties to reduce vulnerability to mechanical stress.
We are also studying how cells in soft biological tissues (e.g., tendon) respond to external, multiaxial mechanical forces and deformations to alter tissue mechanical properties in vitro. Combining data from this study with our ultrasound-based characterization of in vivo tissue deformations during exercises and other activities, we can identify exercise interventions most likely to reverse pathology-associated mechanical changes.
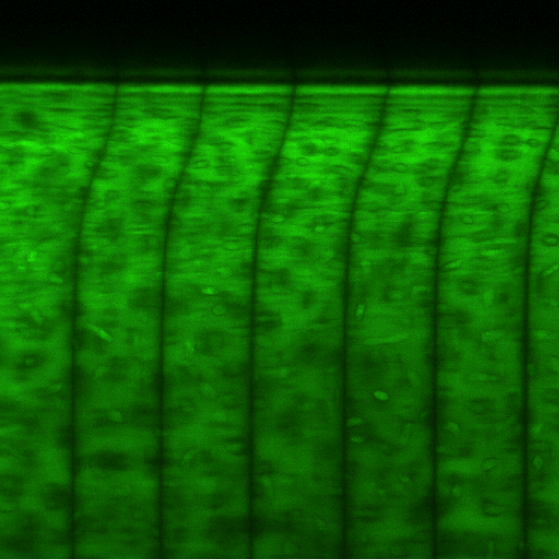
Confocal micrograph of articular cartilage under dynamic shear loading. The black stripes are photobleached lines used as markers to track time-dependent tissue deformation.
Research Interests
- Soft tissue biomechanics, surgical biomechanics, viscoelasticity and poroelasticity, novel imaging techniques to characterize soft tissue properties
