Mukaibo lab comes up with simple way to separate microalgae used for biofuel, other products
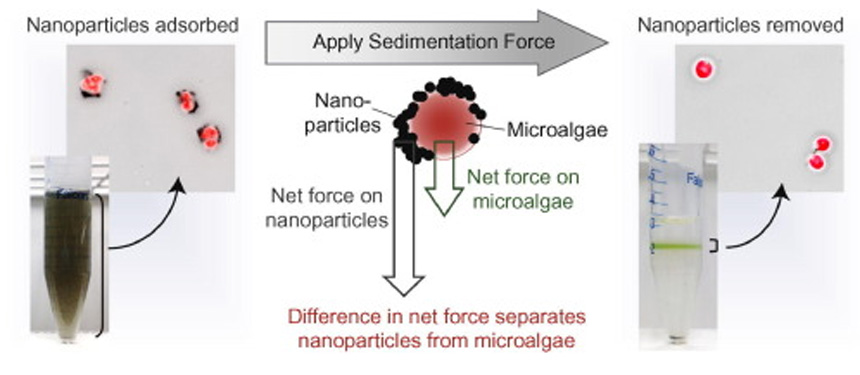
Illustration from the lab of Hitomi Mukaibo, assistant professor of chemical engineering, summarizes sedimentation-induced detachment of magnetite nanoparticles from microalgal flocs.
One of the challenges in using microalgae to produce biofuel, drugs or chemicals is how to separate out the “good stuff,” for example the reusable microalgae, from the slurry-like biomass that remains after harvesting.
One approach, called flocculation, uses nanoparticles as glues that clump the tiny algae together in “flocs”, which make it easier to retrieve them. Magnetic nanoparticles are particularly attractive because these flocs can be collected from the solution by simple force of magnetic field.
One major challenge here is to separate the algae from the nanoparticles, so that both the algae and nanoparticles can be reused. Unfortunately, complex steps or toxic chemicals are typically used to separate the microalgae from the nanoparticles.
The lab of Hitomi Mukaibo, assistant professor of chemical engineering, has come up with a relatively simple method that uses the competing forces of sedimentation on one hand, and flotation and viscosity (or drag) on the other, to pull the algae and nanoparticles apart -- without the extra cost or toxic chemicals involved in other approaches.
This “offers significant opportunity for purifying microalgal biomass after nanoparticle-flocculation-based harvesting and (for) decreasing the cost of microalgal biotechnology,” the researchers note in their article in Renewable Energy Global Innovations. Lead author Shofu Matsuda, a PhD student at Waseda University, worked on this project at Mukaibo lab during the summer of 2015 with co-author graduate students Andrew Durney and Lijie He under the supervision of Mukaibo, the corresponding author.
Currently, two approaches are used to separate the microalgae from the nanoparticles after flocculation:
1. Adding chemicals to dissolve the nanoparticles into the surrounding solution so they can be precipitated out. This can be toxic to the algae and the environment.
2. Orchestrating complex changes to the chemical properties at the algae/nanoparticle interface so the nanoparticles start to slip off from the algae on their own.
Mukaibo says her lab’s approach is “much easier,” capitalizing on the fact that, because of their different sizes, the microalgae and the nanoparticles experience different degrees of buoyancy and drag force.
“The nanoparticles, because they are so small, have less buoyancy and less drag force. So they are more susceptible to an applied sedimentation force (such as centrifuge or magnetic force),” Mukaibo explained. “The algae are bigger and want to float, and experience larger drag forces that prevent them from moving through the fluid,” Mukaibo said. ”The nanoparticles want to go down faster, but the algae can’t keep up. So if you have a high enough force that pulls the particles down, a significant tensile force develops between the fast-moving nanoparticles and the slow moving algae. This eventually separates the two apart.”
The lab used wild type Chlamydomonas Reinhardtii microalgae, bound to magnetite nanoparticles to form flocs. A solution containing these algal flocs were layered onto Percoll®, a commercially available, high-density/viscosity solution. The researchers experimented to find the right adjustments in the viscosity and density of the Percoll® solution. When sedimentation force was applied to the container, the nanoparticles separated from the algae and sank to the bottom of the Percoll® layer, while the algae remained at the interface of the two layers.
Mukaibo believes the technique could be easily applied. “For anyone using centrifuge or magnetic force for separation, all they need to do is make the fluid that the algae is suspended in a little more viscous or a little more dense, and it should be able to separate very effectively.“
The article, which can be read here , was identified as a Key Scientific Article contributing to the excellence in Energy research at Renewable Energy Global Innovations.
