Team
- Amanda Strenk
- Milenia Rojas
- Tatenda Jonga
- Laura Harder
Mentor
Melodie Lawton
Abstract
The purpose of this project is to create functional electrospun nanofiber patches for oromucosal ibuprofen (IBU) delivery. The patches were created using a blend of polycaprolactone (PCL) and polyvinylpyrrolidone (PVP). It was discovered that the optimal blend for drug delivery and fiber formation was a 70:30 ratio of PCL:PVP.
Introduction
Electrospun nanofibers for oral transmucosal drug delivery is an alternative to drug delivery systems such as tablets, needles, and capsules. Electrospun nanofibers increase drug solubility, bioavailability, and manipulate the rate and site of delivery. The purpose of this project was to build a functional low cost electrospinner to create nanofiber patches of PVP/PCL blends infused with IBU to study drug delivery release rates in phosphate buffered saline (PBS). This study can benefit pediatric, geriatric, and other patients with swallowing and digestion problems who are in need of a rapid-release drug.
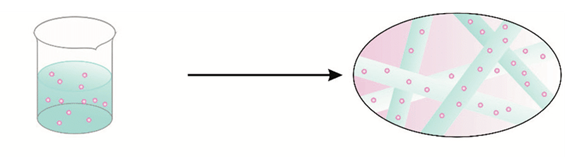
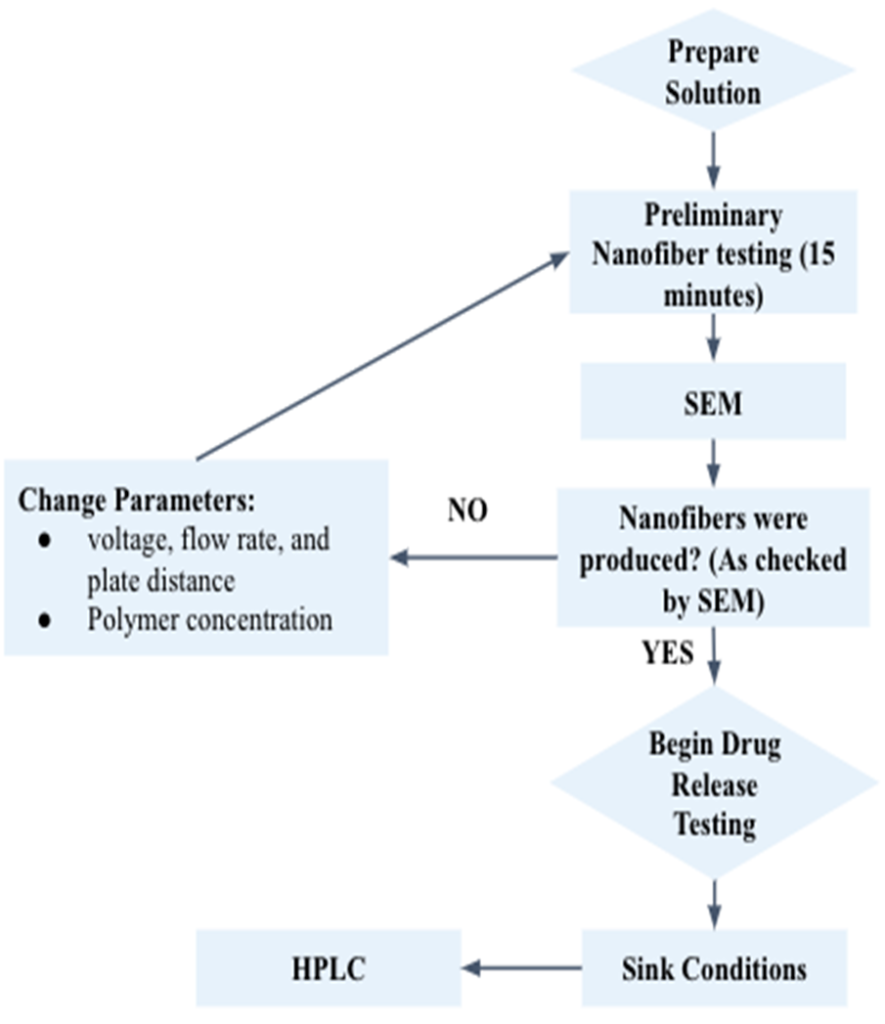
Methods
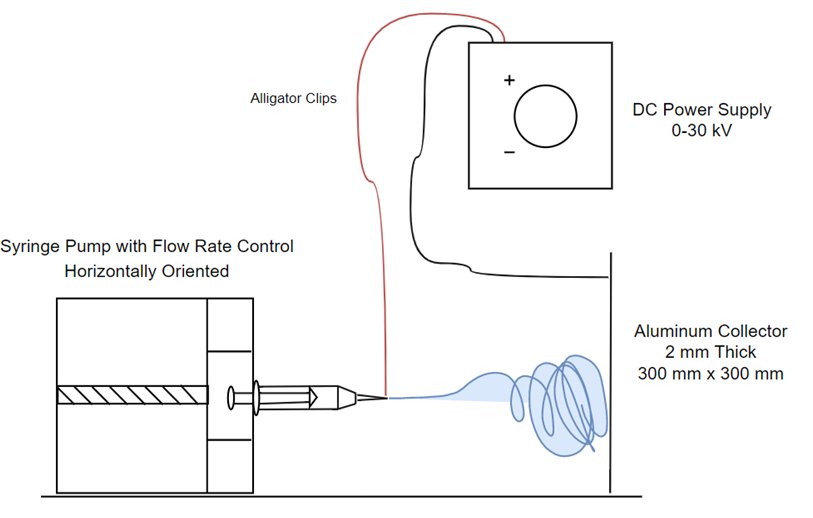
Drug Release Model [2]
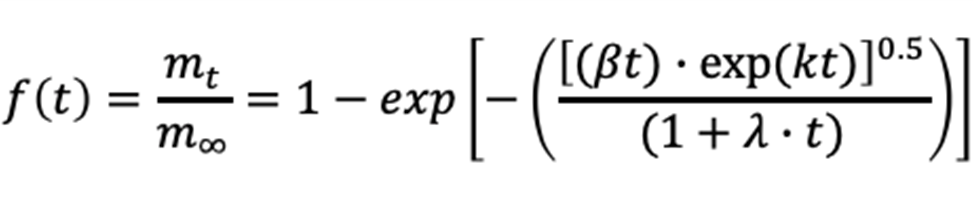
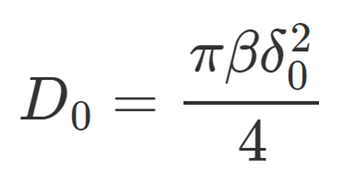
Model Assumes:
• Degradation and Swelling
• Polymer Matrix
• Time-Dependent Diffusion Coefficient
• Time-Dependent Thickness

Hypothesis
1. Films release less IBU than nanofiber patches due to the smaller surface
2. Pure PCL patches will reach peak IBU release after about 4 hours, whereas pure PVP patches will reach peak release after about 3 minutes
3. A PCL/PVP blend will have a shorter release time than PCL on its own
4. Higher PCL wt. % in PCL:PVP blends → Larger nanofiber diameters
5. Larger nanofiber diameters → Lower maximum percent IBU release
Results/Discussion
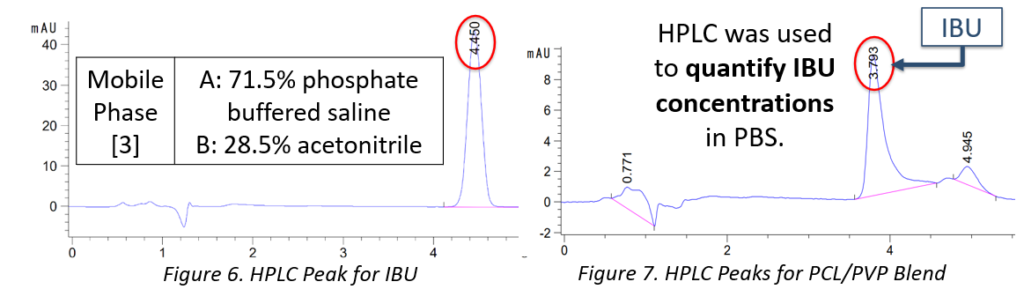
HPLC
SEM Nanofiber Analysis

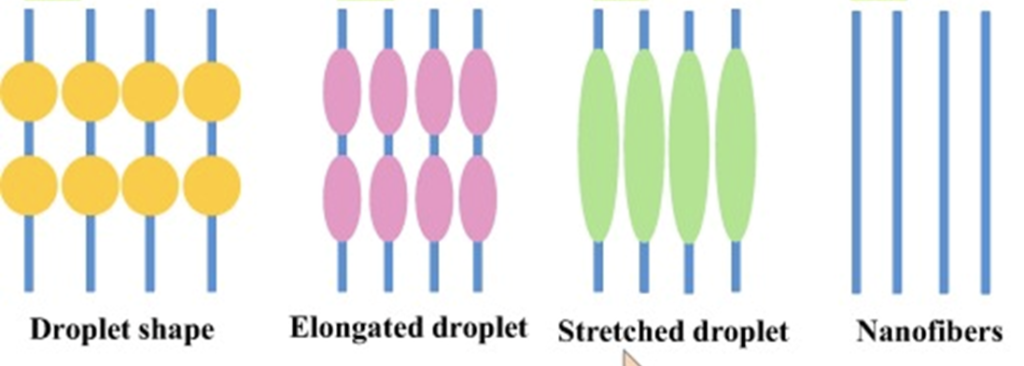
ImageJ software was used to determine the nanofiber average diameter and standard deviation (n = 10). Optimal conditions for electrospinning are chosen based on the smallest standard deviation and no beading/clumping in the nanofibers.
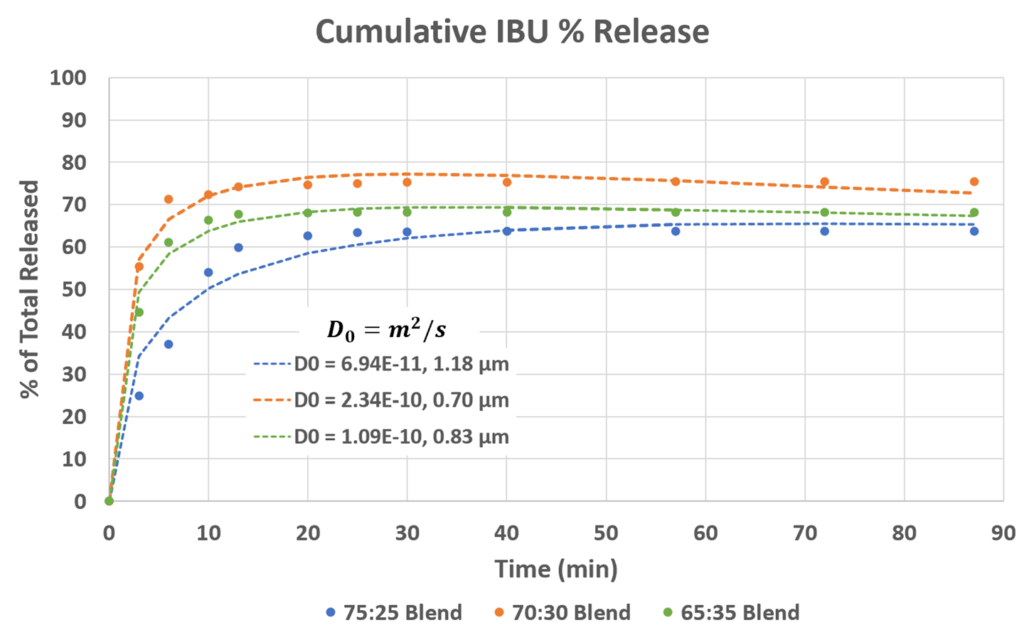
Drug Release Profiles
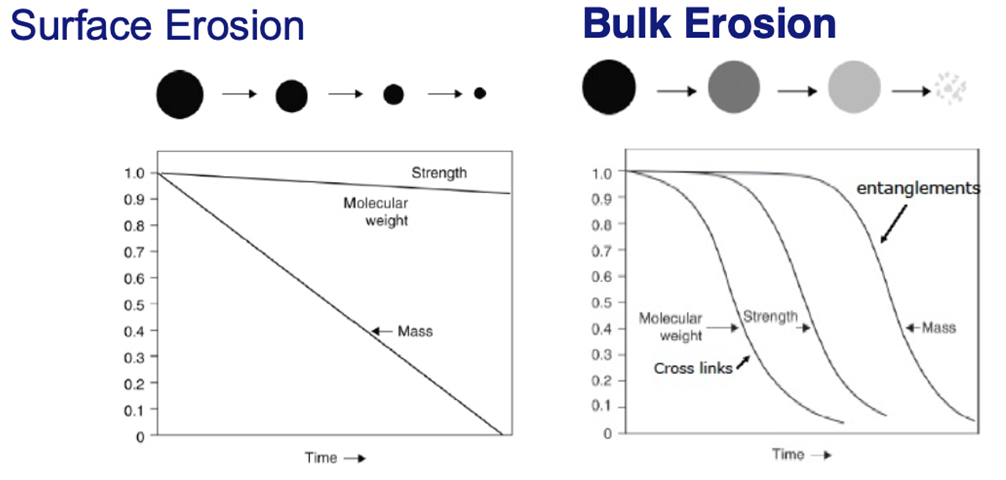
Each blend (PCL:PVP) exhibits an initial burst release from the surface and then diffusion from the bulk. However, there is some IBU trapped on the inner scaffold of the patches as 100% release is not reached.
Overall:
Smaller nanofiber diameter → Larger diffusion coefficient → Maximum % release
Mass Erosion
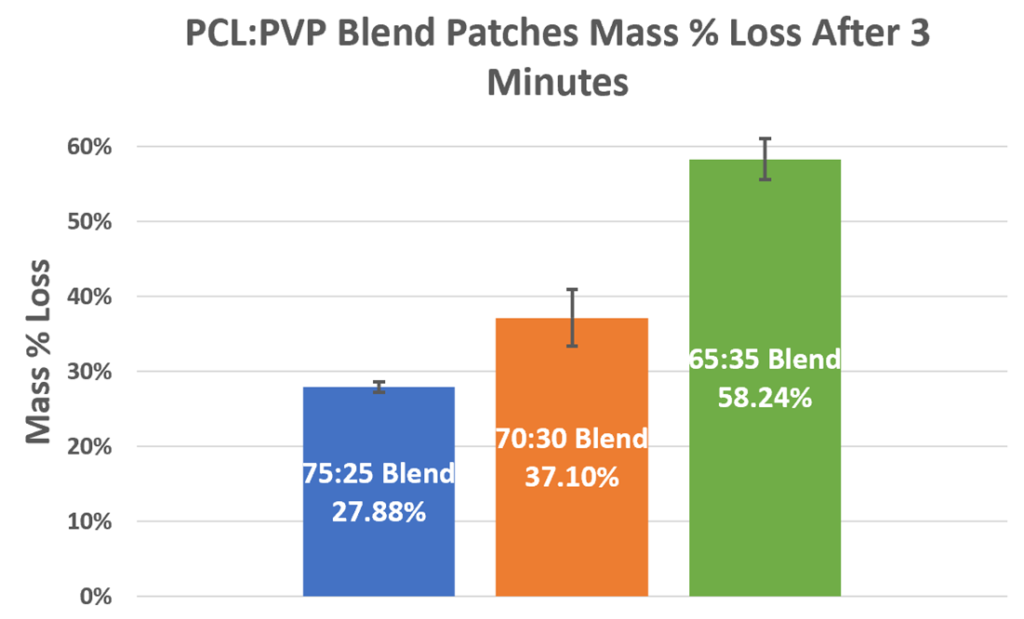


Due to PVP’s hydrophilic nature, it dissolves immediately in the PBS media. Thus, within the first three minutes of submersion in PBS, all of the PVP leaves the patches.
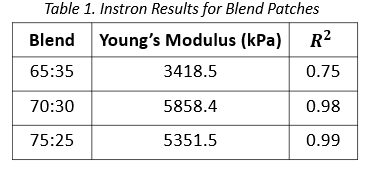
Instron

Final Design

Conclusion
Hypotheses Update:
1. Films released less IBU than nanofiber patches
2. PCL patches reached peak release in about 40 minutes, PVP released immediately
3. PCL/PVP blends had quicker release than pure PCL by about 20 minutes
4. Very little correlation between PCL wt.% and fiber diameter
5. Larger Fiber diameters resulted in a lower maximum release
Overall:
• PCL and PVP blends were electrospinnable
• 70:30 PCL to PVP blend saw the largest maximum release
• Patch thickness contributes to the diffusion coefficient
• The blend patches exhibit surface erosion
• Release kinetics follow the theoretical model
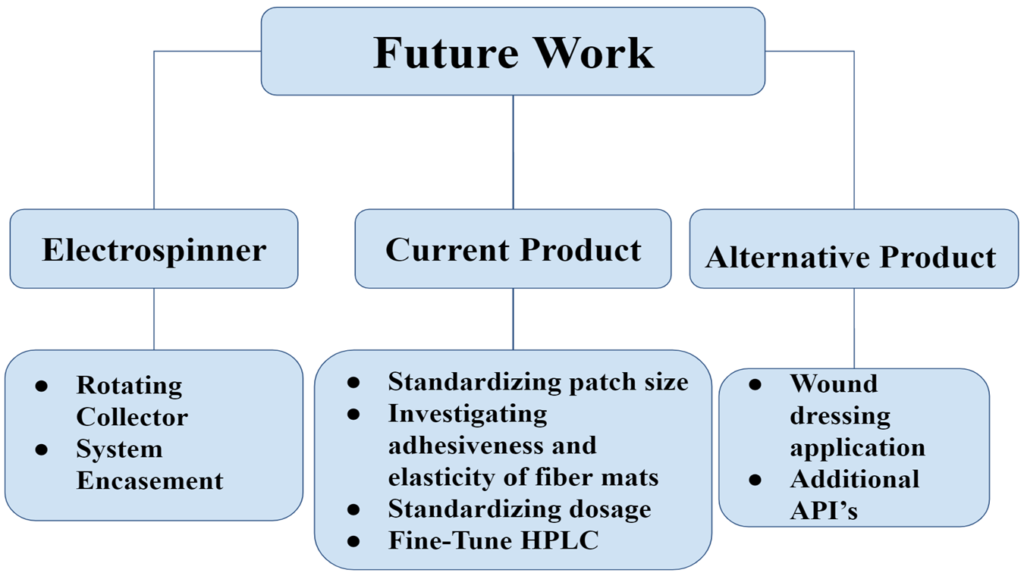
Future Work & Recommendations
Acknowledgements
Our team would like to thank our advisor/sponsor Prof. Lawton for all of her support and advice throughout the semester. We would also like to thank the laboratory staff: Clair Cunningham, Jeff Lefler, Prof. Juba, Prof. Kelley, Prof. Benoit, Mason Garlatti, and Mark Volkin for their technical support and guidance.
References
[1] Torres-Martinez, Erick José, et al. “A Summary of Electrospun Nanofibers as Drug Delivery System: Drugs Loaded and Biopolymers Used as Matrices.” Current Drug Delivery, vol. 15, no. 10, 2018, pp. 1360–1374., https://doi.org/10.2174/1567201815666180723114326. [2] Awad, H. (2022, April 6). Controlled Release Lectures 1-3 [Lecture Notes]. Department of Biomedical Engineering, University of Rochester. [3] Luboz, V., Promayon, E., & Payan, Y. (2014). Linear elastic properties of the facial soft tissues using an aspiration device: Towards patient specific characterization. Annals of Biomedical Engineering, 42(11), 2369–2378. https://doi.org/10.1007/s10439-014-1098-1